The Mole Project:
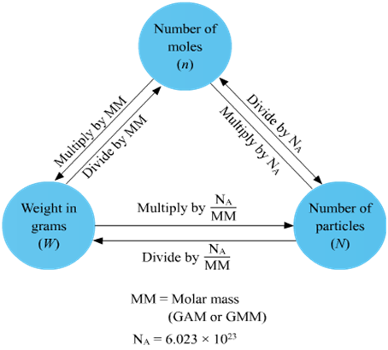
Part 1: -Avogadro's number is the number of atoms in one mole of an element. One mole of oxygen has a mass of 16 grams (the atomic weight).
One mole of hydrogen has a mass of 1 gram (its atomic weight).
Avogadro number says if you have one mole of something, it has 6.022*10^23 of atoms in it.
So, theres 6.022*10^23 atoms in 16 grams of oxygen, just as there is 6.022*10^23 atoms also in 1 gram of hydrogen.
Part 1: -Avogadro's number is the number of atoms in one mole of an element. One mole of oxygen has a mass of 16 grams (the atomic weight).
One mole of hydrogen has a mass of 1 gram (its atomic weight).
Avogadro number says if you have one mole of something, it has 6.022*10^23 of atoms in it.
So, theres 6.022*10^23 atoms in 16 grams of oxygen, just as there is 6.022*10^23 atoms also in 1 gram of hydrogen.
ANZAC BISCUITS
0.63 MOLES OF WATER HAS BEEN REDUCED TO 0.45 MOLES OF WATER.
TIMES 0.68 PER PRODUCT
0.63 MOLES OF WATER HAS BEEN REDUCED TO 0.45 MOLES OF WATER.
TIMES 0.68 PER PRODUCT
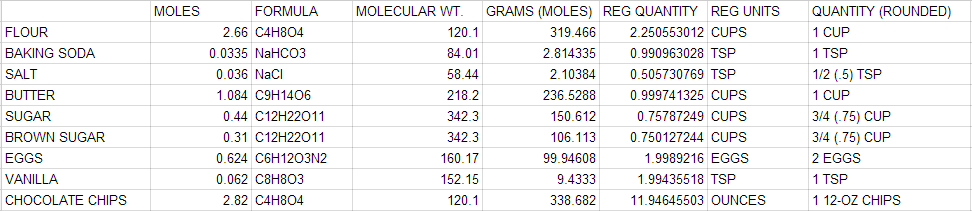
- 2.66 mol Flour: 1.80 mol
- 1.77 mol Rolled Oats: 1.208 mol
- 4.58 mol Brown Sugar: 3.114 mol
- 1.48 mol Shredded Coconut: 1.0064 mol
- .95 mol Almonds: 0.646 mol
- .96 mol Cranberries: 0.6528 mol
- 1.19 mol Butter: 0.8092 mol
- .89 mol Honey: 0.6052 mol
- .63 mol Water: 0.43 moL
OATMEAL CHOCLATE COOKIES
0.72 MOLES OF ROLLED OATS HAS BEEN REDUCED TO 0.50 OF ROLLED OATS.
TIMES .694 PER PRODUCT
0.72 MOLES OF ROLLED OATS HAS BEEN REDUCED TO 0.50 OF ROLLED OATS.
TIMES .694 PER PRODUCT
- 4.26 mol Flower: 2.95 mol
- 0.72 mol Rolled Oats: 0.5 mol
- 3.78 mol Butter: 2.62 mol
- 2 mol Eggs: 1.3 mol
- 0.76 mol Walnuts: .527 mol
- 2.5 mol Sugar: 1.908 mol
- 3.8 mol Chocolate Chips: 2.63 mol
- 0.25 mol Baking soda: 0.1735 mol
- 0.2 mol Vanilla: 0.1388 mol
CHOCLATE CHIP COOKIES3,8 MOLES OF CHOCLATE CHIPS HAS BEEN REDUCED TO 2.5 MOLES OF CHOCLATE CHIPS.
TIMES 0.657 PER PRODUCT
TIMES 0.657 PER PRODUCT
- 1.06 mol Cocoa Powder: 0.696 mol
- 3.78 mol Butter: 2.48 mol
- 0.76 mol Eggs: 0.499 mol
- 0.76 mol Walnuts: .499 mol
- 3.35 mol Sugar: 2.2 mol
- 3.66 mol Brown Sugar: 2.404 mol
- 3.8 mol Chocolate Chips: 2.49 mol
- .02 mol Baking Soda: 2.4 mol
- .25 mol Vanilla: 0.164 mol
CHOCLATE CHIP COOKIES3,8 MOLES OF CHOCLATE CHIPS HAS BEEN REDUCED TO 2.5 MOLES OF CHOCLATE CHIPS.
TIMES 0.657 PER PRODUCT
TIMES 0.657 PER PRODUCT
- 1.06 mol Cocoa Powder: 0.696 mol
- 3.78 mol Butter: 2.48 mol
- 0.76 mol Eggs: 0.499 mol
- 0.76 mol Walnuts: .499 mol
- 3.35 mol Sugar: 2.2 mol
- 3.66 mol Brown Sugar: 2.404 mol
- 3.8 mol Chocolate Chips: 2.49 mol
- .02 mol Baking Soda: 2.4 mol
- .25 mol Vanilla: 0.164 mol
1 sandwich= 1 cheese + 2 slices of bread
13 bread slices / 2 slices per sandwich = ~5 sandwiches w/ 5 cheeses worth.
5 sandwiches + cheese is the limiting reagent.
13 bread slices / 2 slices per sandwich = ~5 sandwiches w/ 5 cheeses worth.
5 sandwiches + cheese is the limiting reagent.
Number of sandwiches per ingredient:
Patties: 8
Buns: 6
Cheese: 7
Tomato: 9
Lettuce: 8
Onion: 9
Theoretical number is metion next the ingredient.
The Buns give you the lowest number theoretical yield which makes it the limiting reagent, 6.
Patties: 8
Buns: 6
Cheese: 7
Tomato: 9
Lettuce: 8
Onion: 9
Theoretical number is metion next the ingredient.
The Buns give you the lowest number theoretical yield which makes it the limiting reagent, 6.
1. LiOH + CO2 --> Li2CO3 + H2O
-Balanced: 2LiOH + CO2 --> Li2CO3 + H2O
-Theoretical yield of each reagent: LiOH: molar mass 23.95
1g/ 23.95 = 0.0417 mol
output of water : ratio of water to reagent: 1:2
theoretical: 0.0208 mol
theoretical output of CO2:
molar mass: 44.01 1g/44.01=0.022 mol
ratio w water & reagent 1:1
theoretical output: 0.0227 mol
LiOH is the limiting reagent.
91.8% yield.
2. Zn + AgNO2 --> Ag + Zn(NO3)2
-Balanced: Zn + 3AgNO2 --> 3Ag + Zn(NO3)2
Zn: Molar mass (65.39)
1g/153.87 = 0.00649 mol
Ratio of Ag to AgNO2 --> 3:3 Theoretical output: 0.00649 mol
AgNO2 is the limiting reactant
Theoretical yield: 42.69%
-Balanced: 2LiOH + CO2 --> Li2CO3 + H2O
-Theoretical yield of each reagent: LiOH: molar mass 23.95
1g/ 23.95 = 0.0417 mol
output of water : ratio of water to reagent: 1:2
theoretical: 0.0208 mol
theoretical output of CO2:
molar mass: 44.01 1g/44.01=0.022 mol
ratio w water & reagent 1:1
theoretical output: 0.0227 mol
LiOH is the limiting reagent.
91.8% yield.
2. Zn + AgNO2 --> Ag + Zn(NO3)2
-Balanced: Zn + 3AgNO2 --> 3Ag + Zn(NO3)2
Zn: Molar mass (65.39)
1g/153.87 = 0.00649 mol
Ratio of Ag to AgNO2 --> 3:3 Theoretical output: 0.00649 mol
AgNO2 is the limiting reactant
Theoretical yield: 42.69%