Redox Reaction
Redox Reaction: Redox (reduction-oxidation) reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The oxidation numbers are part of redox as if any of the atoms in the reactants are different from the oxidation number for the same atoms on the product side, electron transfer must have occurred, and you have a redox reaction.

A real life redox reaction would be iron rust, that happens using combustion that occurs when chemical bonds are broken and new bonds are formed. Combustion occurs in a fast pace when it involved rusting, it's an oxidation-reduction.
A very important tip when it comes to solving a redox problem is OILRIG.
Oxygen
Is
Loss
Reduction
Is
Gain
Oxygen
Is
Loss
Reduction
Is
Gain
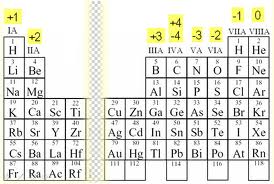
Finding the oxidation number for the following...
1.) NH4+1
- the oxidation number for N would be -3
2.) HMnO4
- the oxidation number for Mn is -7
3.) H2SO3
- the oxidation number of S is -4
- the oxidation number for N would be -3
2.) HMnO4
- the oxidation number for Mn is -7
3.) H2SO3
- the oxidation number of S is -4
1 C10H8 + 7 O2 --> 5CO2 + 4 H2O
There would be a redox reaction.
Oxygen would be reduced by -2.
Carbon would be oxidized.
2 Rb + 3F2 --> 2RbF
no redox.
2 K + 1 Zn (NO3)2 --> 1Zn + 2KNO3
redox reaction.
Zn is reduced.
K is oxidated.
There would be a redox reaction.
Oxygen would be reduced by -2.
Carbon would be oxidized.
2 Rb + 3F2 --> 2RbF
no redox.
2 K + 1 Zn (NO3)2 --> 1Zn + 2KNO3
redox reaction.
Zn is reduced.
K is oxidated.