objective 1
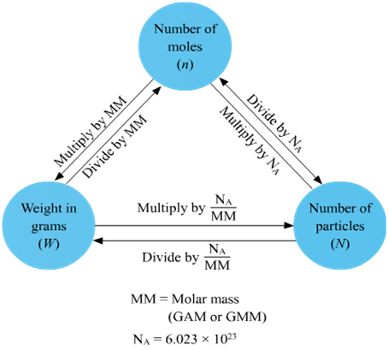
avogadro's number related to moles
Avogadro's number: 6.022 x 10^23
Avogadro's number is the number of atoms in one mole of an element.
Avogadro's number is the number of atoms in one mole of an element.
- One mole of oxygen has a mass of 16 grams (the atomic weight).
- One mole of hydrogen has a mass of 1 gram (its atomic weight).
- Avogadro number says if you have one mole of something, it has 6.022*10^23 of atoms in it.
- There is 6.022*10^23 atoms in 16 grams of oxygen, just as there is 6.022*10^23 atoms also in 1 gram of hydrogen.
Objective 2
ANZAC BISCUITS
0.63 MOLES OF WATER HAS BEEN REDUCED TO 0.45 MOLES OF WATER.
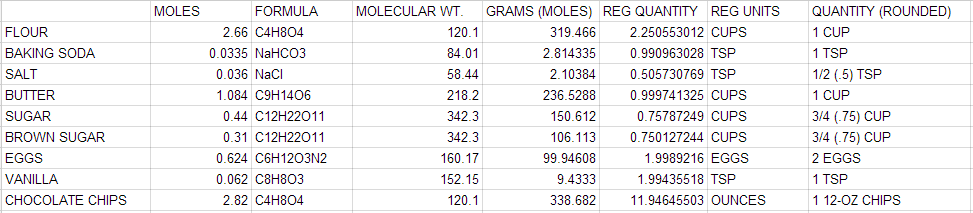
- 2.66 mol, Flour: 1.80 mol
- 1.77 mol, Rolled Oats: 1.208 mol
- 4.58 mol,Brown Sugar: 3.114 mol
- 1.48 mol, Shredded Coconut: 1.0064 mol
- .95 mol, Almonds: 0.646 mol
- .96 mol, Cranberries: 0.6528 mol
- 1.19 mol, Butter: 0.8092 mol
- .89 mol, Honey: 0.6052 mol
- .63 mol, Water: 0.43 mol
OATMEAL CHOColate cookies
0.72 MOLES OF ROLLED OATS HAS BEEN REDUCED TO 0.50 OF ROLLED OATS.
- 4.26 mol, Flower: 2.95 mol
- 0.72 mol, Rolled Oats: 0.5 mol
- 3.78 mol, Butter: 2.62 mol
- 2 mol, Eggs: 1.3 mol
- 0.76 mol, Walnuts: .527 mol
- 2.5 mol, Sugar: 1.908 mol
- 3.8 mol, Chocolate Chips: 2.63 mol
- 0.25 mol, Baking soda: 0.1735 mol
- 0.2 mol, Vanilla: 0.1388 mol
CHOColate chip cookies
3,8 MOLES OF CHOCLATE CHIPS HAS BEEN REDUCED TO 2.5 MOLES OF CHOCOLATE CHIP COOKIES.
- 1.06 mol, Cocoa Powder: 0.696 mol
- 3.78 mol, Butter: 2.48 mol
- 0.76 mol, Eggs: 0.499 mol
- 0.76 mol, Walnuts: .499 mol
- 3.35 mol, Sugar: 2.2 mol
- 3.66 mol, Brown Sugar: 2.404 mol
- 3.8 mol, Chocolate Chips: 2.49 mol
- .02 mol, Baking Soda: 2.4 mol
- .25 mol, Vanilla: 0.164 mol
DROP SUGAR COOKIES WITH WALNUTS
5.33 MOLES OF FLOUR HAS BEEN REDUCED TO 4.33 MOLES OF FLOUR.
- 5.33 mol, Flour: 4.32 mol
- 2.35 mol, Butter: 1.908 mol
- .48 mol, Eggs: 0.389 mol
- .96 mol, Walnuts: 0.779 mol
- 4.19 mol, Sugar: 3.4 mol
- .25 mol, Baking Soda: .203 mol
- .31 mol, Vanilla: 0.251 mol
Objective 3
Problem solving
1. You want to make 6 grilled cheese sandwiches (use the equation below, Bd = bread and Ch = Cheese) and you have 13 slices of bread and 5 slices of cheese. How may grilled cheese sandwich can you make? And what is the limiting reagent?
2Bd + Ch→Bd2Ch
Bd = 13 Ch = 5
You have 5 slices of cheeses there for you can only have 5 grilled cheese sadndwiches. Your cheese is the limmiting reagent.
2Bd + Ch→Bd2Ch
Bd = 13 Ch = 5
You have 5 slices of cheeses there for you can only have 5 grilled cheese sadndwiches. Your cheese is the limmiting reagent.
2. You are having some friends over for dinner you want to make 8 cheese burgers, you have 8 hamburger patties, 12 bums, 7 slices of cheese, 18 slices of tomato, 16 pieces of lettuce and 9 slices of onion. Use the equation below (where buns = B, hamburger patty = H, cheese = Ch, tomatoes = T lettuce = L and onion = O) to determine how many cheese burgers you can make (what’s your theoretical yield?) and what the limiting reagent is? Hint you may need to balance the equation first.
B+ H+ Ch+ 2T+ 2L+O → BHChT2L2O
# of sandwiches per ingredient (theoretical #)
patties: 8
buns: 6
cheese:7
tomato: 9
lettuce: 8
onion:9
buns=lowest theoretical yield || limiting reagent=6
B+ H+ Ch+ 2T+ 2L+O → BHChT2L2O
# of sandwiches per ingredient (theoretical #)
patties: 8
buns: 6
cheese:7
tomato: 9
lettuce: 8
onion:9
buns=lowest theoretical yield || limiting reagent=6
3. Balance the chemical equations below. Find the limiting reagent and the theoretical yield if there is one gram of each reagent. After each step, write a sentence or two explaining what you did.
LiOH+CO2 → LiCO3+H2O
Zn+AgNO2 → Ag+Zn(NO3)2
1. 2LiOH + CO2 --> Li2CO3 + H2O
Theoretical yield of each reagent: LiOH: molar mass 23.95
1g/ 23.95 = 0.0417 mol
output of water : ratio of water to reagent: 1:2
theoretical: 0.0208 mol
theoretical output of CO2:
molar mass: 44.01 1g/44.01=0.022 mol
ratio w water & reagent 1:1
theoretical output: 0.0227 mol
LiOH is the limiting reagent.
91.8% yield.
2. Zn + 3AgNO2 --> 3Ag + Zn(NO3)2
Zn: Molar mass (65.39)
1g/153.87 = 0.00649 mol
Ratio of Ag to AgNO2 --> 3:3 Theoretical output: 0.00649 mol
AgNO2 is the limiting reactant
Theoretical yield: 42.69%
LiOH+CO2 → LiCO3+H2O
Zn+AgNO2 → Ag+Zn(NO3)2
1. 2LiOH + CO2 --> Li2CO3 + H2O
Theoretical yield of each reagent: LiOH: molar mass 23.95
1g/ 23.95 = 0.0417 mol
output of water : ratio of water to reagent: 1:2
theoretical: 0.0208 mol
theoretical output of CO2:
molar mass: 44.01 1g/44.01=0.022 mol
ratio w water & reagent 1:1
theoretical output: 0.0227 mol
LiOH is the limiting reagent.
91.8% yield.
2. Zn + 3AgNO2 --> 3Ag + Zn(NO3)2
Zn: Molar mass (65.39)
1g/153.87 = 0.00649 mol
Ratio of Ag to AgNO2 --> 3:3 Theoretical output: 0.00649 mol
AgNO2 is the limiting reactant
Theoretical yield: 42.69%