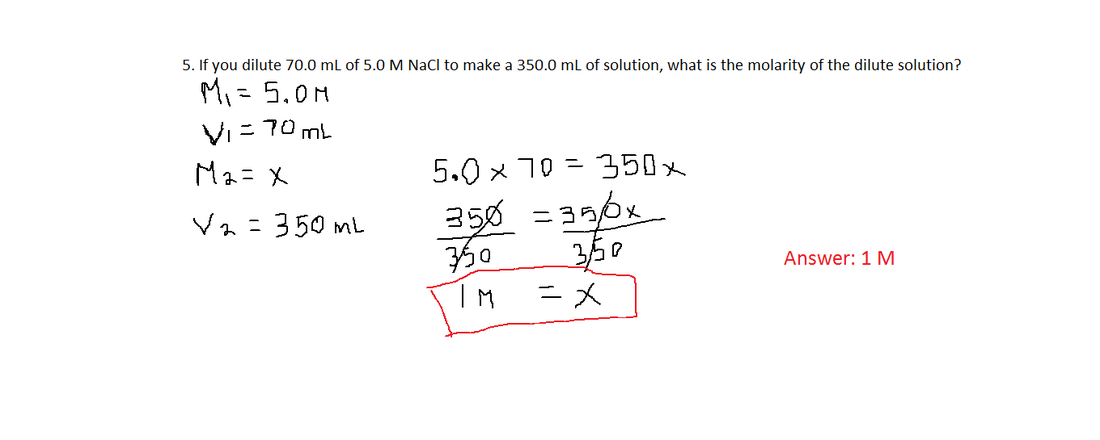sOLUTIONS
What is a solution?
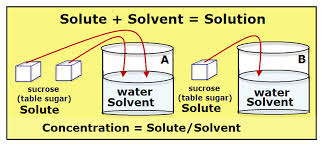
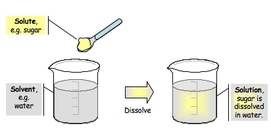
Homogeneous mixture composed of only one phase in such a mixture, as solute is a substance dissolved in another substance, known as a solvent.
Homogeneous mixture composed of only one phase in such a mixture, as solute is a substance dissolved in another substance, known as a solvent.
Parts of the solution:
Solute: The substance being dissolved.
Solvent: The substance doing the dissolving or the substance present in the largest amount. Water is the universal solvent.
- In the pictures we know our two substances are sugar and water. As we know, water is the universal solvent, therefore it is the solvent and the sugar is the solute. Together they are the solution.
Water - Solvent
Sugar- Solute
Solute: The substance being dissolved.
Solvent: The substance doing the dissolving or the substance present in the largest amount. Water is the universal solvent.
- In the pictures we know our two substances are sugar and water. As we know, water is the universal solvent, therefore it is the solvent and the sugar is the solute. Together they are the solution.
Water - Solvent
Sugar- Solute
Three factors that affect the rate of solutions
1. Solids become more soluble as the temperature increases.
2. Gases become less soluble as the temperature increases.
- Pressure has no effect on solids and liquids.
3. Gases become more soluble as the pressure increases.
2. Gases become less soluble as the temperature increases.
- Pressure has no effect on solids and liquids.
3. Gases become more soluble as the pressure increases.
MOlarity
Molarity Formula: moles of solute / liters of solution
Solving following problems:
MOLality
Molality Formula: moles of solute / kilogram of solvent
Solving following problem:
Dilution
Dilution Formula: M1V1 = M2V2
Solving following problems:
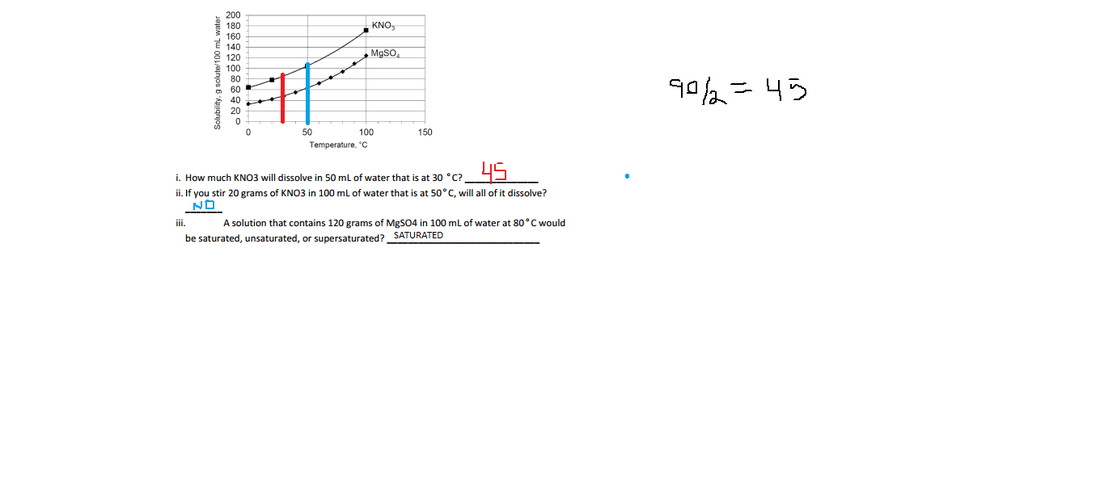
Solubility
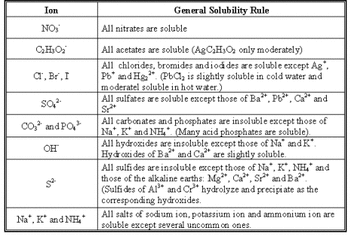
Solubility Rules