Boyles law

INFORMATION ABOUT ROBERT BOYLE
Robert Boyle was an Irish natural philosopher, chemist, and inventor during the 17th century . Born in January 25,1627 in Republic Ireland and died in December 30,1691. Robert brought many advances to chemistry, however he is mostly known for his important discovery, the Boyle’s Law, which he decided to publish it in 1662. Today Robert is credited for being one of the earliest modern chemists. Robert was also one of the few credited for being one of the pioneers of the modern experimental scientific method. The law in which the relationship between the absolute pressure and volume of a gas is measured, in which temperature is kept constant within a system, was given the name of Boyles.
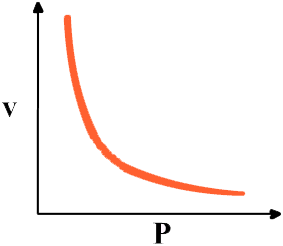
Boyle's law states Boyle's law states that if the temperature of a gas is kept constant, the pressure if the gas is inversely proportional to the volume.
P1V1 = P2V2
Robert Boyle was an Irish natural philosopher, chemist, and inventor during the 17th century . Born in January 25,1627 in Republic Ireland and died in December 30,1691. Robert brought many advances to chemistry, however he is mostly known for his important discovery, the Boyle’s Law, which he decided to publish it in 1662. Today Robert is credited for being one of the earliest modern chemists. Robert was also one of the few credited for being one of the pioneers of the modern experimental scientific method. The law in which the relationship between the absolute pressure and volume of a gas is measured, in which temperature is kept constant within a system, was given the name of Boyles.
Boyle's law states Boyle's law states that if the temperature of a gas is kept constant, the pressure if the gas is inversely proportional to the volume.
P1V1 = P2V2
Examples of boyle's law in real life
This gas law can be seen used on daily bases.
When the plunger of the syringe is pulled back the volume of the syringe container increases, that way it decreases the pressure inside the syringe. Since the same amount of gas is now spread over a greater volume. To be able to balance this effect of low pressure, air or blood is sucked in through the needle, that way balancing the pressure inside and outside the syringe.
- Change of pressure in a syringe
When the plunger of the syringe is pulled back the volume of the syringe container increases, that way it decreases the pressure inside the syringe. Since the same amount of gas is now spread over a greater volume. To be able to balance this effect of low pressure, air or blood is sucked in through the needle, that way balancing the pressure inside and outside the syringe.

- Increase in size of bubbles as they rise to the surface
- Scuba diving, we have all see when scuba divers breathing under water create bubbles. These bubbles grow as they rise to the surface. The deeper a scuba diver goes in the sea, means more pressure. Therefore as the bubbles rise to the surface the pressure decreases and accordingly the volume increases.
P1V1 = P2V2
- P is pressure, measured in atm.
- V is volume, measured in liters (L)
Pressure and volume are both included in the ideal gas law, PV = nRT. PV = nRT can be broken down as follows:
P = pressure (measured in atm).
V = volume (measured in liters).
n = moles.
R = the universal gas constant (.0821)
T = temperature (measured in Kelvin).
P = pressure (measured in atm).
V = volume (measured in liters).
n = moles.
R = the universal gas constant (.0821)
T = temperature (measured in Kelvin).
Boyle's gas law example
5.00 L of a gas is at 1.08 atm. What pressure is obtained when the volume is 10.0 L?
- Using the formula P1V1 = P2V2
- 1.08 atm x 5.00 L = (x) 10.0 L
- X = 1.08 atm x 5.00 L / 10.0 L
- Answer will be: 0.54 atm
Gas law problems
1. A gas occupies 12.3 liters at a pressure of 40.0 mm Hg. What is the volume when the pressure is increased to 60.0 mm Hg?
2. If a gas at 25.0 °C occupies 3.60 liters at a pressure of 1.00 atm, what will be its volume at a pressure of 2.50 atm?
3. To what pressure must a gas be compressed in order to get into a 3.00 cubic foot tank the entire weight of a gas that occupies 400.0 cu. ft. at standard pressure?
4. A gas occupies 1.56 L at 1.00 atm. What will be the volume of this gas if the pressure becomes 3.00 atm?
5. A gas occupies 11.2 liters at 0.860 atm. What is the pressure if the volume becomes 15.0 L?
2. If a gas at 25.0 °C occupies 3.60 liters at a pressure of 1.00 atm, what will be its volume at a pressure of 2.50 atm?
3. To what pressure must a gas be compressed in order to get into a 3.00 cubic foot tank the entire weight of a gas that occupies 400.0 cu. ft. at standard pressure?
4. A gas occupies 1.56 L at 1.00 atm. What will be the volume of this gas if the pressure becomes 3.00 atm?
5. A gas occupies 11.2 liters at 0.860 atm. What is the pressure if the volume becomes 15.0 L?
Answers to Problems
1. 40.0 mm Hg x 12.3 liters = 60.0 mm Hg (x); x = 8.20 L
2. 1.00 atm x 3.60 liters = 2.50 atm (x); x = 1.44 L
3. 400.0 cu. ft x 1.00 atm = 3.00 cubic ft (x) ; x = 133 atm
4. 1.56 L x 1.00 atm = 3.00 atm (x); 0.520 L
5. 11.2 liters x 0.860 atm = (x) 15.0 L; x = 0.642 atm
2. 1.00 atm x 3.60 liters = 2.50 atm (x); x = 1.44 L
3. 400.0 cu. ft x 1.00 atm = 3.00 cubic ft (x) ; x = 133 atm
4. 1.56 L x 1.00 atm = 3.00 atm (x); 0.520 L
5. 11.2 liters x 0.860 atm = (x) 15.0 L; x = 0.642 atm
By leslie Banda
sources:
http://www.citycollegiate.com/gaslaws.htm
http://sciencehelp.blog.com/2013/04/29/boyles-law-sample-problem/
http://ucluscubaclub.com/gallery/thailand-gallery/hannah-diving-bubbles/
http://antoine.frostburg.edu/chem/senese/101/gases/faq/everyday-gas-laws.shtml
http://www.citycollegiate.com/gaslaws.htm
http://sciencehelp.blog.com/2013/04/29/boyles-law-sample-problem/
http://ucluscubaclub.com/gallery/thailand-gallery/hannah-diving-bubbles/
http://antoine.frostburg.edu/chem/senese/101/gases/faq/everyday-gas-laws.shtml