Types of REACTIONS
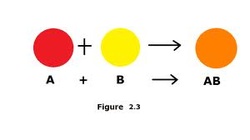
Synthesis (Combination) - Two elements as reactants to make ONE product.
- Metal elements must go first.
- Metal elements must go first.
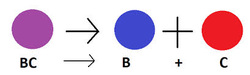
Decomposition - ONE reactant breaks apart into its element.
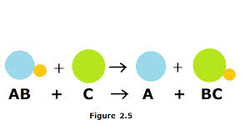
Single Replacement - Characterized by having an element and compound as reactants and the products are another element and another compound.

Activity series of metals in Aqueous Solution. Using this is helpful when it comes to single replacement.
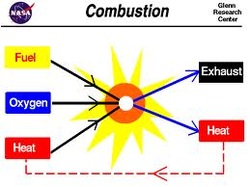
Combustion - A hydrocarbon reacts with oxygen to produce carbon dioxide and water.
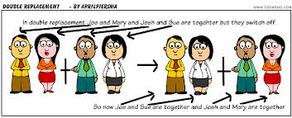
Double Replacement - is a chemical reaction where two reactant ionic compounds exchange ions to form two new product compounds with the same ions.
Solved.
Zn(s) + S8(s) --> ZnS(s)
- Synthesis.
Al2S3(s) --> Al(s) + S8(s)
- Decomposition
Mg(OH)2 (aq) + H2SO4 (aq) --> MgSO4 + H2O (I)
- Double replacement
C12H22O11(g) + O2(g) --> CO2(g) + H2O (g)
- Combustion
H2SO4(aq) +Fe(s) --> H2(g) + FeSO4(aq)
- single replacement
Zn(s) + S8(s) --> ZnS(s)
- Synthesis.
Al2S3(s) --> Al(s) + S8(s)
- Decomposition
Mg(OH)2 (aq) + H2SO4 (aq) --> MgSO4 + H2O (I)
- Double replacement
C12H22O11(g) + O2(g) --> CO2(g) + H2O (g)
- Combustion
H2SO4(aq) +Fe(s) --> H2(g) + FeSO4(aq)
- single replacement

