The law of conservation of mass
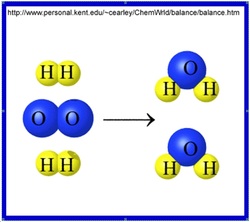
The Law of Conservation of Mass states that matter cannot be created nor destroyed in an ordinary chemical reaction. No atoms are gained or lost in a reaction; they are just rearranged. It stays balanced.
When solving a chemical equation and using the law of conservation we must always be able to have the same amount of atoms.They can be split into two parts or more but it will stay balanced. In other words the chemical equation can be rearranged.
Chemical equations applying the Law of Conservation:
1.) K2CO3 --> K2O + CO2
1 K2CO3 --> 1 K2O + 1 CO2
- chemical equation already balanced. Broken into two parts.
1.) K2CO3 --> K2O + CO2
1 K2CO3 --> 1 K2O + 1 CO2
- chemical equation already balanced. Broken into two parts.
2.) K3PO4 + HCl --> KCl + H3PO4
1 K3PO4 + 3 HCl --> 3 KCl + 1 H3PO4
K3PO4 + H3Cl3 --> K3Cl3 + H3PO4
1 K3PO4 + 3 HCl --> 3 KCl + 1 H3PO4
K3PO4 + H3Cl3 --> K3Cl3 + H3PO4
3.) Na + HNO3 --> NaNO3 + H2
2 Na + 2 HNO3 --> 2 NaNO3 + 1 H2
Na2 + H2N2O6 --> Na2N2O6 + H2
2 Na + 2 HNO3 --> 2 NaNO3 + 1 H2
Na2 + H2N2O6 --> Na2N2O6 + H2
4.) Sil4 + Mg --> Si + Mgl2
1 Sil4 + 2 Mg --> 1 Si + 2 Mgl2
Sil4 + Mg2 --> Si + Mg2l4
1 Sil4 + 2 Mg --> 1 Si + 2 Mgl2
Sil4 + Mg2 --> Si + Mg2l4
5.) Ca(ClO3)2 --> CaCl2 + O2
1 Ca(ClO3)2 --> 1 CaCl2 + 3 O2
CaCl2O6 --> CaCl2 + O6
1 Ca(ClO3)2 --> 1 CaCl2 + 3 O2
CaCl2O6 --> CaCl2 + O6
